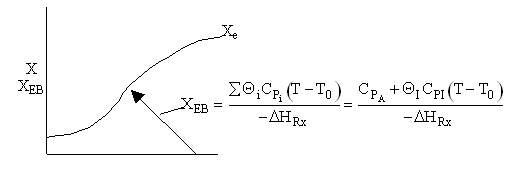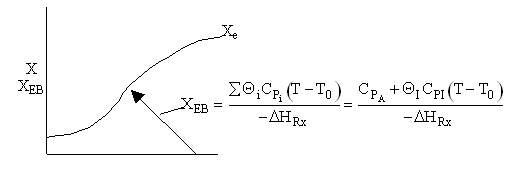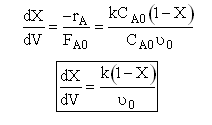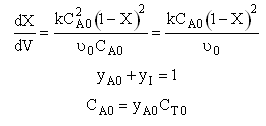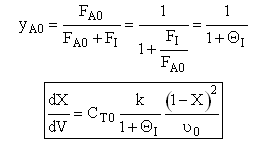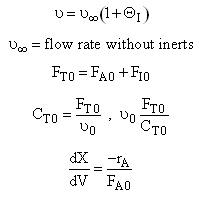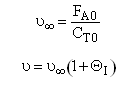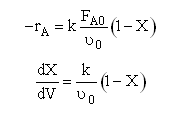Chapter 8 Example
Adiabatic Endothermic Reaction
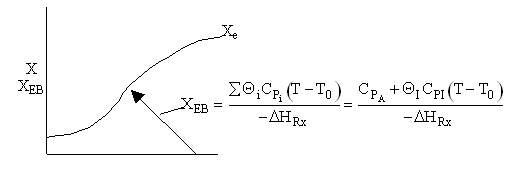
PFR Adiabatic
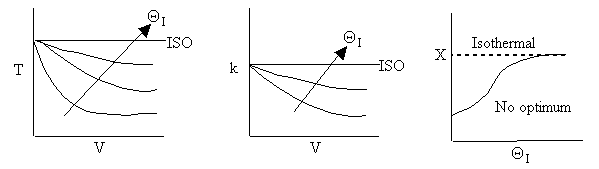
Case A Fix FT0, CT0 and therefore υ0 is consistent as ΘI is varied
- Irreversible A → B
Liquid Phase
A. First order
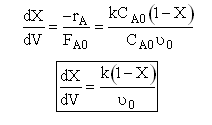
First Order irreversible
B. Second order
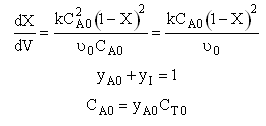
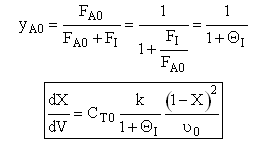
Second Order Irreversible
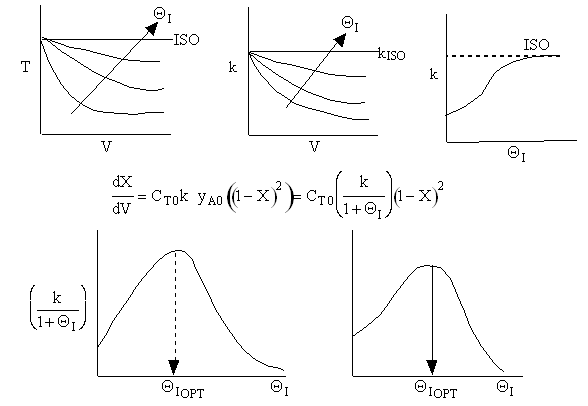
Case B For Fixed FA0, CT0 and Variable FI0 therefore variable υ0
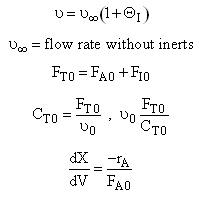
Let ν∞ be the volumetric flow with pure A.
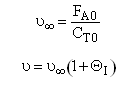
First order reaction
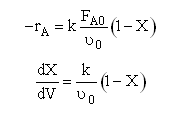
Now as inerts are added to the volumetic flow rate ν0 will increase
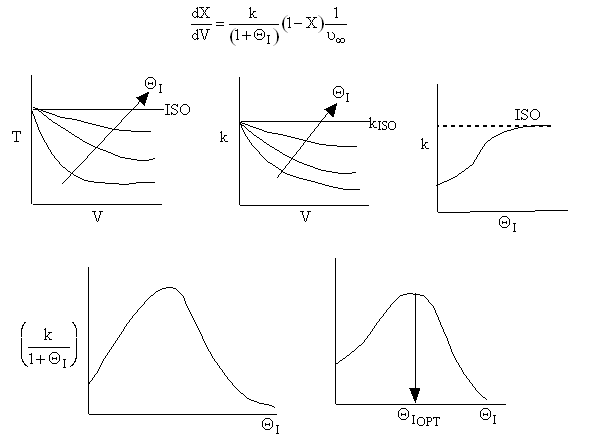
The same will hold true for a second order reaction for fixed FA0
Back to Chapter 8