An extremely brief introduction to computational quantum chemistry
In this section, we provide a very brief background for the computational
tools to be used in this module, which are based on quantum chemistry. Because
quantum theory is not the primary concern of this exercise, important themes
in quantum chemistry are addressed in a broad manner. Quantum theory holds
that matter has both particle- and wavelike properties. For large particles,
the wavelike properties of matter are nearly impossible to detect. However,
for small particles—such as electrons—the particle-wave duality
must be addressed when writing equations, including energy balance equations.
The quantum mechanical energy balance (also known as the Schrödinger
equation) for an electron can be written as follows:
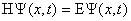 (1)
(1)
In this equation, E is the total energy of the electron, 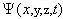 psi is the wavefunction for the electron, and H is the Hamiltonian operator
that describes the kinetic and potential energy of the electron. In other
words, the Schrödinger equation is a simple statement that the total
energy is equal to the kinetic plus potential energies for a wavelike particle.
The Hamiltonian operator for a single electron can be written as:
psi is the wavefunction for the electron, and H is the Hamiltonian operator
that describes the kinetic and potential energy of the electron. In other
words, the Schrödinger equation is a simple statement that the total
energy is equal to the kinetic plus potential energies for a wavelike particle.
The Hamiltonian operator for a single electron can be written as:
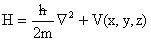 (2)
(2)
Where V (x,y,z) = 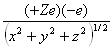 (3)
(3)
Z is the atomic number, e is the charge associated with an electron, and
x,y, and z are spatial components.
And 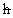 is
Planck’s constant divided by two Pi, m is the mass of an electron,
is
Planck’s constant divided by two Pi, m is the mass of an electron, 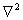 is the Laplacian
operator, and V(x,y,z) is the potential energy term, which depends on
the electrostatic interactions of the electron with nuclei (attractive interactions)
and other electrons (repulsive interactions) in the system.
is the Laplacian
operator, and V(x,y,z) is the potential energy term, which depends on
the electrostatic interactions of the electron with nuclei (attractive interactions)
and other electrons (repulsive interactions) in the system.
As we will see from the examples in this module, by finding the total energy
of molecular systems by solving Schrödinger equations, we can compute
thermodynamic and kinetic quantities that are important in chemical reaction
engineering (CRE). We start with a solution for the simplest possible molecule
– the hydrogen atom, with a single nucleus and single electron –
and then show how the results from this solution can be used to address more
complex (and relevant) chemical systems.
The Hydrogen-Like Atom
The Schrödinger equation can only be solved analytically for a “hydrogen-like”
atom, i.e., an atom with one electron and one nucleus. The Schrödinger
equation is a special type of equation known as an eigenvalue equation, which
some of you may have encountered in higher-level mathematics courses. Eigenvalue
problems have a number of special characteristics. One of these is that they
have an infinite number of solutions for both the eigenvalues ( )
and the associated eigenfunctions(
)
and the associated eigenfunctions( ).
The value n (an integer) is known in quantum mechanics as the principle quantum
number. The fact that there are an infinite number of solutions for the energy
of our molecule may seem confusing at first, but we will soon see what these
solutions for different values of n represent.
).
The value n (an integer) is known in quantum mechanics as the principle quantum
number. The fact that there are an infinite number of solutions for the energy
of our molecule may seem confusing at first, but we will soon see what these
solutions for different values of n represent.
The Schrödinger equation—like many other partial differential equations—can
be solved using a solution method known as separation of variables subject
to the boundary condition that the energy of the system is zero when the electron
and nucleus are infinitely far away from each other. We will not go through
the details of the solution method here; that method is described in a number
of references (click
here for references). Instead, we will discuss the form of the solutions
to the Schrödinger equation for the hydrogen-like atom.
The lowest energy solution for n=1 describes the state in which the system
is most probable to exist; this is also known as the ground state. For n=2
(the next-lowest energy solution) there are four solutions that have equivalent
energies; for n=3 (the third-lowest energy solution), the number of energetically
equivalent solutions rises to 9. Although the physical meaning of the total
energies  for the system are relatively easy to understand, the meaning of the wavefunction
is not. To gain insights into what the wavefunctions actually are, we will
consider the quantity:
for the system are relatively easy to understand, the meaning of the wavefunction
is not. To gain insights into what the wavefunctions actually are, we will
consider the quantity:
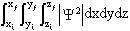 (4)
(4)
Which is a Cartesian coordinate triple integral over all of the space that
the electron could occupy. It can be shown that this quantity describes the
probability that the electron will be located in the volume of space between
the endpoints labeled i (initial) and f (final). To make this more clear,
one can plot the  probability distribution as a function of position for each of our solutions.
probability distribution as a function of position for each of our solutions.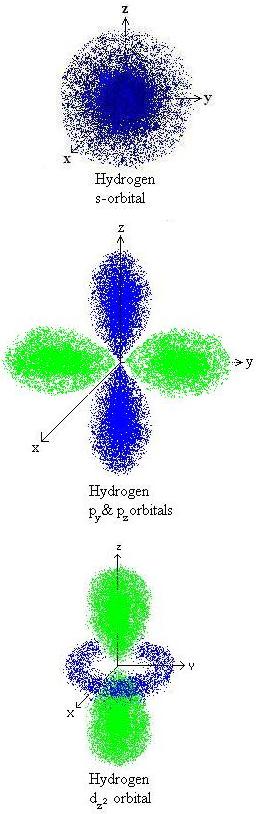
We see that our eigenfunctions psi 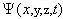 actually describe the electronic orbitals of the hydrogen atoms. We can recognize
that the lowest energy solution (for n=1) corresponds to the 1s orbital:which
is the orbital that we know should be occupied by an electron in the ground
state for an H atom. This is the region in space in which the electron is
most probable to be present. For n=2, there are four energetically degenerate
wavefunctions that represent the 2s and 2px, 2py, and 2pz orbitals; these
are orbitals which are less energetically favorable for occupation by the
electron, as we know from basic chemistry. Furthermore, the nine energetically
degenerate 3s, 3p, and 3d orbitals are still higherin energy. We could continue
going to higher energy solutions describing the 4s, p, d, and f orbitals,
etc., but this information is usually not of practical interest to us. That’s
because we’re interested in the ground state of the molecule,
the one that preferentially exists in nature (and in our chemical application).
So we’re interested in the lowest-energy solution to the Schrödinger
equation, the one in which the wavefunction describes a hydrogen 1s orbital.
In the exercises in this module, we will show how to use this ground state
energy to evaluate thermodynamic properties.
actually describe the electronic orbitals of the hydrogen atoms. We can recognize
that the lowest energy solution (for n=1) corresponds to the 1s orbital:which
is the orbital that we know should be occupied by an electron in the ground
state for an H atom. This is the region in space in which the electron is
most probable to be present. For n=2, there are four energetically degenerate
wavefunctions that represent the 2s and 2px, 2py, and 2pz orbitals; these
are orbitals which are less energetically favorable for occupation by the
electron, as we know from basic chemistry. Furthermore, the nine energetically
degenerate 3s, 3p, and 3d orbitals are still higherin energy. We could continue
going to higher energy solutions describing the 4s, p, d, and f orbitals,
etc., but this information is usually not of practical interest to us. That’s
because we’re interested in the ground state of the molecule,
the one that preferentially exists in nature (and in our chemical application).
So we’re interested in the lowest-energy solution to the Schrödinger
equation, the one in which the wavefunction describes a hydrogen 1s orbital.
In the exercises in this module, we will show how to use this ground state
energy to evaluate thermodynamic properties.
Larger atoms and molecules
There is no analytical solution to the Schrödinger equation for systems
containing multiple nuclei and electrons. This difficulty is caused by the
fact that repulsive interactions with multiple electrons greatly complicate
the solution to the Schrödinger equation. Therefore, we must use an approximate,
numerical solution. The best way to do this is to assume that the wavefunctions
can be based on the same basic shapes identified for the H atom; that is,
that they should describe orbitals that look like 1s, 2s, 2p, etc., and (in
the case of chemical bonds formed between atoms) linear combinations of these
orbitals. We will therefore construct our numerical solution for the eigenvalues
and eigenfunctions using a basis set of orbital shapes (i.e., s, p, d, and
f wave functions) to describe the wavefunction for each electron:
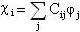 (5)
(5)
where  describes the different functions that approximate electronic orbitals, and
describes the different functions that approximate electronic orbitals, and
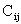 are fitting
coefficients that are varied in the course of our numerical solution of the
Schrödinger equation. In other words, our solution method will involve
varying the coefficients
are fitting
coefficients that are varied in the course of our numerical solution of the
Schrödinger equation. In other words, our solution method will involve
varying the coefficients 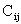 until a minimum energy (ground state) solution is found. The energy minimum
that is found is not always the absolute global minimum for the molecule,
because an molecule can have many local energy minima. A principle known as
the variational principle dictates that the lowest energy solution will be
closest to the true energy of the molecular system. The larger the basis
set size—i.e., the more fitting coefficients we have at our disposal
for the solution—the closer to the true ground state energy our solution
will be. However, larger basis sets also naturally require longer computational
times, because there will be more terms to include in the calculation. An
infinite basis set – one that includes innumerable different shapes for
the fitting functions – could in principle be used to compute an exact
molecular energy. But this calculation would take an infinite amount of time!
We typically use the smallest basis set size possible that generates results
in reasonably good agreement with experiments.
until a minimum energy (ground state) solution is found. The energy minimum
that is found is not always the absolute global minimum for the molecule,
because an molecule can have many local energy minima. A principle known as
the variational principle dictates that the lowest energy solution will be
closest to the true energy of the molecular system. The larger the basis
set size—i.e., the more fitting coefficients we have at our disposal
for the solution—the closer to the true ground state energy our solution
will be. However, larger basis sets also naturally require longer computational
times, because there will be more terms to include in the calculation. An
infinite basis set – one that includes innumerable different shapes for
the fitting functions – could in principle be used to compute an exact
molecular energy. But this calculation would take an infinite amount of time!
We typically use the smallest basis set size possible that generates results
in reasonably good agreement with experiments.
There are a number of ways to use these basis sets in solving the Schrödinger
equation; we will deal only with some of the more commonly used methods here.
One of the earliest methods for treating many-electron systems is based on
the Hartree-Fock (HF) approximation. The discovery of the HF method was central
to the field of quantum chemistry, and it forms the heart of many of the more
accurate computational methods today. In the HF method, the energy of each
electron is solved for individually, using a modified version of the Schrödinger
equation called the Hartree-Fock equation:
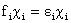 (6)
(6)
This equation is solved for each electron i, where epsilon (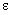 )
is the energy of the electron analogous to E in the Schrödinger equation,
chi (
)
is the energy of the electron analogous to E in the Schrödinger equation,
chi (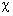 )
its electronic orbital analogous to
)
its electronic orbital analogous to  ,
and ( f ) is the so-called Fock operator analogous to H. The Fock operator
has the form:
,
and ( f ) is the so-called Fock operator analogous to H. The Fock operator
has the form:
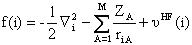 (7)
(7)
Where 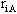 is
the radial distance be tween the ith electron and the Ath nucleus,
is
the radial distance be tween the ith electron and the Ath nucleus, 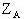 is the atomic number of the Ath nucleus, and
is the atomic number of the Ath nucleus, and 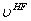 is the average potential felt by the ith electron from the presence of the
other electrons present in the system.
is the average potential felt by the ith electron from the presence of the
other electrons present in the system.
Solving the complete system of HF equations (as discussed below), one can
determine energies for each electron, and sum these together with nuclear
nuclear repulsion energies to obtain the total energy of the molecule. The
key to the HF approximation is the use of the term 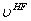 ,
which describes the average electronic charge “felt” by the ith
electron; this average is used to compute the effect of electrostatic interactions
between electrons on the shapes and energies of the individual orbitals. Taking
the average effect of the electrostatic interactions is far less cumbersome
to calculate as opposed to calculating the electrostatic effect of the system
(the molecule) on each individual electron.
,
which describes the average electronic charge “felt” by the ith
electron; this average is used to compute the effect of electrostatic interactions
between electrons on the shapes and energies of the individual orbitals. Taking
the average effect of the electrostatic interactions is far less cumbersome
to calculate as opposed to calculating the electrostatic effect of the system
(the molecule) on each individual electron.
Solution of the HF equations is accomplished by an algorithm known as the
self-consistent field (SCF) procedure. The computation basically proceeds
as follows:
- An initial guess for the values of the basis set coefficients,
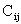 ,
is made.
,
is made.
- The values of
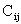 are used to construct the electronic orbitals
are used to construct the electronic orbitals  through a linear combination of single electron orbitals.
through a linear combination of single electron orbitals.
- Utilizing the electronic orbitals constructed in step 2, which tells
you on average the location of the electrons, allows one to determine
the average electrostatic interaction (
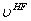 )
between the electron in question and the remaining electrons in the system.
This electrostatic interaction is calcuated for each electron and then
averaged to more accurately represent the electron-electron interactions
in the molecule.
)
between the electron in question and the remaining electrons in the system.
This electrostatic interaction is calcuated for each electron and then
averaged to more accurately represent the electron-electron interactions
in the molecule.
- Once
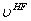 is known, the Hartree-Fock equations can be solved to determine new electronic
orbitals and their associated energies.
is known, the Hartree-Fock equations can be solved to determine new electronic
orbitals and their associated energies.
- The electronic energies can be summed to calculated the total energy
of the system. If the energy is sufficiently converged with respect to
the previous SCF cycle, we’re done. If not, return to step 3 and
continue to cycle until the energy value converges to a stable (minimum)
value.
In many of the programs that are used for these computations, you will be
able to watch the progress of the SCF procedure as the calculation progresses.
Although the HF method has produced many valuable results, it turns out that
significant errors can be induced by treating repulsive forces between electrons
in an average sense, rather than treating them individually. We refer to the
difference between the HF energy and the true energy of the system (molecule),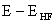 ,
as the correlation energy. There are many methods which attempt to improve
the accuracy of computed energies using different methods of accounting for
the electron correlation. We list several of these below, along with a very
brief description of the basis for each method.
,
as the correlation energy. There are many methods which attempt to improve
the accuracy of computed energies using different methods of accounting for
the electron correlation. We list several of these below, along with a very
brief description of the basis for each method.
- Density Functional Theory (DFT). In DFT, the correlation
energy is treated as a function of the electronic density, which itself
is a function of position. In other words, the correlation can be calculated
for a given electronic charge distribution in the molecular system. A
number of functionals have been devised for the correlation energy; these
functionals vary in their effectiveness for given applications. Treating
the correlation energy as a functional of the density greatly improves
computational accuracy without a significant cost in terms of computer
time. In fact, because other terms in the Schrodinger equation can be
treated as functionals of the density, DFT calculations sometimes run
faster than HF computations. For this reason, DFT
is widely used for studying complex systems in heterogeneous catalysis,
electronic thin films deposition, etc.
- Perturbation theories. These theories account for the
correlation energy in terms of perturbations to the original eigenfunction
and eigenvalue solutions. These perturbations are treated as series expansions
of the original eigenfunctions. Two of the most popular perturbation methods
are referred to as Moller-Plessett calculations, which are commonly based
on 2nd-order or 4th-order expansions and referred to as MP2 and MP4, respectively.
MP4 is more accurate, but requires longer computation times.
- Coupled cluster theories. Coupled cluster theories are a method of electronic structure
thoery for predicting molecular properties. The basic ideas of CC theories includes cluster expansion, cluster
operators and size extensivity. The theory can be furthur categorized into aa formal theory and a pratical
theory. The CC equations are quadratic in the ampitudes and must be solved iteratively. CC is correct to the
infinite order for single and double excitations and is correct to the 4th order for quadruple excitations.
- Configuration interaction. Configuration interaction (CI)
methods hypothetically can calculate exactly the correlation energy in
the limit of an infinite basis set (i.e., an infinite number of fitting
parameters). Meaning that the calculation of the total system energy is
exact. The price that is paid for this exactness, of course, is an extremely
long computation time – full CI is not yet practical for most systems
of chemical engineering interest. In CI, the exact wave function is represented
as a linear combination of the HF ground state wave function plus excited
state wave functions. Similar to perturbation methods, the more excited
states that are included in the calculation, the more accurate (and lengthy)
the calculation.

(1)
![]() psi is the wavefunction for the electron, and H is the Hamiltonian operator
that describes the kinetic and potential energy of the electron. In other
words, the Schrödinger equation is a simple statement that the total
energy is equal to the kinetic plus potential energies for a wavelike particle.
The Hamiltonian operator for a single electron can be written as:
psi is the wavefunction for the electron, and H is the Hamiltonian operator
that describes the kinetic and potential energy of the electron. In other
words, the Schrödinger equation is a simple statement that the total
energy is equal to the kinetic plus potential energies for a wavelike particle.
The Hamiltonian operator for a single electron can be written as:(2)
![]() (3)
(3) ![]() is
Planck’s constant divided by two Pi, m is the mass of an electron,
is
Planck’s constant divided by two Pi, m is the mass of an electron, ![]() is the Laplacian
operator, and V(x,y,z) is the potential energy term, which depends on
the electrostatic interactions of the electron with nuclei (attractive interactions)
and other electrons (repulsive interactions) in the system.
is the Laplacian
operator, and V(x,y,z) is the potential energy term, which depends on
the electrostatic interactions of the electron with nuclei (attractive interactions)
and other electrons (repulsive interactions) in the system.